About Concord Biotech
A Global Biotechnology Company
Pioneering Biotech for Sustainable Growth
Concord Biotech Limited is a leading, R&D driven biopharmaceutical company focussed on driving sustainable growth through manufacturing fermentation-based Active Pharmaceutical Ingredients (APIs) and formulations. Concord Biotech leverages its technical expertise and cutting-edge processes to produce high-quality products.
Our commitment to excellence is reinforced through globally regulatory-approved manufacturing facilities that operate in line with the highest standards of quality and compliance, enabling Concord to continually establish new industry benchmarks.
Our commitment to excellence is reinforced through globally regulatory-approved manufacturing facilities that operate in line with the highest standards of quality and compliance, enabling Concord to continually establish new industry benchmarks.
Milestones
Milestones Achieved by
Milestones Achieved by
Concord Biotech
2000-2005
The company has seen significant developments, including acquisition by Mr. Sudhir Vaid, the certification of the Dholka facility for enzyme production, and the first USFDA inspection of the Dholka API manufacturing facility.
2006-2010
The company successfully completed the second USFDA inspection, classifying the API facility as acceptable, and received the drug master file registration for Tacrolimus Hydrate from Japan's PMDA.
2011-2015
The Dholka facility has successfully undergone regulatory inspections by the EU GMP, Government of Upper Bavaria, USFDA, and Japan's PMDA for the foreign manufacturing of non-sterile drugs.
2016-2020
The company established Formulation facility, R & D got recognized by DSIR, secured Helix investment, expanded its critical care business in India, and received board approval for putting up of injectable plant.
2021-2024
The company got listed in August 2023 in BSE and NSE, commenced operations at its second API facility in Limbasi, achieved successful inspections of Dholka facility by EU GMP, PMDA, ANVISA (Brazil) etc.
Milestones
Milestones Achieved by
Concord Biotech

- Acquisition of the Company by Mr. Sudhir Vaid.
- Renaming of the Company to Concord Biotech Limited.
- Expansion of enzyme production capacity.
- Strategic Investment by Rakesh Jhunjhunwala and Rekha Jhunjhunwala in the Company.
- First USFDA Inspection of the Dholka API facility
2000 - 2005

- Second USFDA Inspection of the Dholka API Facility
- Drug Master File (DMF) Registration certificate received for Tacrolimus Hydrate from the Pharmaceuticals and Medical Devices Agency (PMDA), Japan.
2006 - 2010

- First EU GMP Inspection of the manufacturing facility by the Government of Upper Bavaria – Central Medicines Control Bavaria (GMP/GCP) of our Dholka API Facility.
- Initiation of CRAM Services in the areas of New Chemical Entities (NCEs) and Generic APIs.
- Drug Substance Registration certificate received for Mycophenolate Mofetil and Cyclosporine JP from the Japan FDA.
- Two USFDA Inspection and Second EU GMP Inspection of the Dholka facility successfully concluded.
- Accreditation Certificate of foreign drug manufacturing for non-sterile drugs received from the Minister of Health, Labour and Welfare, Japan.
2011- 2015

- Establishment of Formulation Facility at Valthera, Gujarat.
- Recognition of the In-house R&D Formulation Facility at Valthera by the Ministry of Science and Technology, Government of India.
- Strategic Investment received from Helix.
- Two USFDA Inspections at the Valthera formulation facility, both successfully concluded.
- Fifth USFDA Inspection successfully completed for the API facility at Dholka.
- GMP Certification received from the Food and Drugs Control Administration (FDCA) for the Valthera and Dholka units.
- Joint Venture Established in Japan to pursue growing business opportunities.
- Approval of Four ANDAs, expanding product reach in regulated markets.
- Expansion into the Critical Care Segment in India.
2016 - 2020

- Commissioning and Commencement of Operations of the second API facility at Limbasi.
- Third EU GMP Inspection successfully completed for the API facility at Dholka.
- Listing of the Company on BSE and NSE.
- Successful Completion of Multiple International Inspections of the Dholka facility by EU GMP, Japan FDA (PMDA), and ANVISA (Brazil).
2021 - 2024

- Strategic Investment in Palvella Therapeutics, Inc., a US-based company.
- Equity Investment of 26% in Clean Max for sourcing renewable energy at the Dholka API plant.
- Commencement and Commissioning of the Injectable facility at Valthera.
- USFDA Approval for marketing of Teriflunomide Tablets.
- Successful Inspections of API facility at Dholka by USFDA, EU GMP, Korea FDA, and Russian GMP.
- Successful Inspection of formulation unit at Valthera by EU GMP and Saudi FDA
- First Inspection of API facility at Limbasi by EU GMP.
2025

- Acquisition of the Company by Mr. Sudhir Vaid.
- Renaming of the Company to Concord Biotech Limited.
- Expansion of enzyme production capacity.
- Strategic Investment by Rakesh Jhunjhunwala and Rekha Jhunjhunwala in the Company.
- First USFDA Inspection of the Dholka API facility
2000 - 2005

- Second USFDA Inspection of the Dholka API Facility
- Drug Master File (DMF) Registration certificate received for Tacrolimus Hydrate from the Pharmaceuticals and Medical Devices Agency (PMDA), Japan.
2006 - 2010

- First EU GMP Inspection of the manufacturing facility by the Government of Upper Bavaria – Central Medicines Control Bavaria (GMP/GCP) of our Dholka API Facility.
- Initiation of CRAM Services in the areas of New Chemical Entities (NCEs) and Generic APIs.
- Drug Substance Registration certificate received for Mycophenolate Mofetil and Cyclosporine JP from the Japan FDA.
- Two USFDA Inspection and Second EU GMP Inspection of the Dholka facility successfully concluded.
- Accreditation Certificate of foreign drug manufacturing for non-sterile drugs received from the Minister of Health, Labour and Welfare, Japan.
2011 - 2015

- Establishment of Formulation Facility at Valthera, Gujarat.
- Recognition of the In-house R&D Formulation Facility at Valthera by the Ministry of Science and Technology, Government of India.
- Strategic Investment received from Helix.
- Two USFDA Inspections at the Valthera formulation facility, both successfully concluded.
- Fifth USFDA Inspection successfully completed for the API facility at Dholka.
- GMP Certification received from the Food and Drugs Control Administration (FDCA) for the Valthera and Dholka units.
- Joint Venture Established in Japan to pursue growing business opportunities.
- Approval of Four ANDAs, expanding product reach in regulated markets.
- Expansion into the Critical Care Segment in India.
2016 - 2020

- Commissioning and Commencement of Operations of the second API facility at Limbasi.
- Third EU GMP Inspection successfully completed for the API facility at Dholka.
- Listing of the Company on BSE and NSE.
- Successful Completion of Multiple International Inspections of the Dholka facility by EU GMP, Japan FDA (PMDA), and ANVISA (Brazil).
2021 - 2024

- Strategic Investment in Palvella Therapeutics, Inc., a US-based company.
- Equity Investment of 26% in Clean Max for sourcing renewable energy at the Dholka API plant.
2025
Our Vision
Create products through Biotechnology route and Service offering that enrich the lives of people by continually building on our expertise in Biotechnology.
Our Mission
We strive to be a global power in the field of Biotechnology based products through research and development to create sustainable earnings growth and to establish long-term business success. We continue to reinforce our commitment to safety, health, and the environment. We endeavor for an environment where creativity and effectiveness are encouraged with the technologies for the niche market.
Our Shared Values at
Concord Biotech
Driving Continuous Progress
We constantly explore new processes, research, and concepts while adopting cutting-edge technologies, fuels our pursuit of long-term success.
Our Moral Compass
We maintain the highest moral and ethical standards, prioritising honesty in both personal and professional conduct.
Setting Global Standards
We adhere to the highest standards, fulfilling customer expectations and improving their quality of life.
Leading the Way
We recognise the importance of sensitivity, compassion, and kindness towards our people, business associates, and society.
Excellence at Core
We are attaining business excellence from research to final product delivery by harnessing teamwork in a time-bound manner.
Dedicated to Deliver
We perform our duties with exemplary moral responsibility and accountability across all functions with a solution-finding mindset.
Our Foundation
We showcase result-oriented actions both at the individual and team levels, which forms the foundation of our work culture.
Leadership
The Minds Behind
Concord Biotech

Mr. Sudhir Vaid
Chairman and Managing Director of Concord Biotech Limited
Mr. Sudhir Vaid, Chairman and Managing Director of Concord Biotech, is a biotechnologist turned entrepreneur with 50 years of expertise in biopharmaceuticals. After completing his studies in microbiology from Punjab University in 1974, he started his career in biotechnology and moved to different domestic and multinational biotechnology companies till he started his own business in the year 2000. In 2000, he founded Concord Biotech, transforming it into a global leader in fermentation-based biopharmaceuticals. A dedicated philanthropist, he contributes to healthcare, education, and research. He also serves on Gujarat’s biotechnology councils, driving innovation and industry growth.
Mr. Ankur Vaid
JMD & CEO of Concord Biotech Limited
Ankur Vaid, one of the Promoter and CEO of Concord Biotech Limited, holds a B. Tech in Chemical Engineering and holds MBA degree. With over 18 years in the pharmaceutical industry, he has been with the company since 2006, leading key initiatives in research, development, and market strategy. His vision and expertise have been central to the company’s continued growth and success.

About Our Company
Team of Professionals
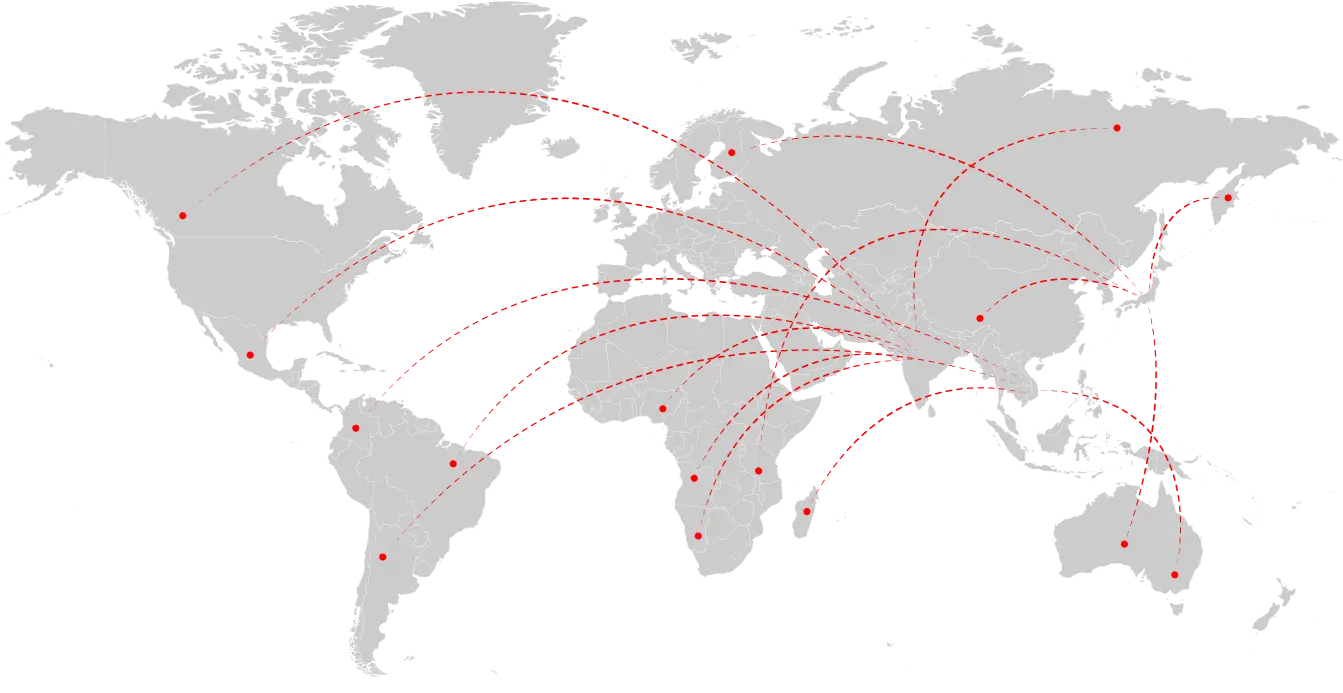
Our Global Presence
A Global Leader in
Biotech Solutions
Concord Biotech Limited is globally known for its products and has a commanding presence in more than 70 countries worldwide with efficient distribution infrastructure in markets like North America, Europe, Japan, Central & Latin America, Africa, Australia, New Zealand, Asia-Pacific as well as CIS and MENA countries, besides significant presence in Indian market. Concord is actively expanding its horizons by partnering with leading Global Pharmaceutical Companies by meeting their product development needs for API’s & Finished Formulations.